Chemical Bonding and Molecular Structure
Lewis approach helps us to write the structures of molecule.
1. Lewis Structure:
Steps For Drawing Lewis Dot Structure:
1. Calculate the total number of valence electrons of all the atoms in the molecule.
2. Choose a central bonding atom from the given molecule.
* Central bonding atom is the least electronegative atom.
* Exceptions: Hydrogen (H) and Fluorine (F) always occupy peripheral position.
3. Draw a single sigma bond (-) between the central bonding atom and each peripheral atom.
4. Draw three unshared electron pairs, called lone-pairs, to complete octet on each peripheral atom except for Hydrogen (H), which
follows duet rule.
* If extra electrons remain, place them on the central bonding atom.
5. Complete octet for the central bonding atom by moving lone pairs from peripheral atoms.
* Atoms with less than four valence electrons like Li, Be, and B have less than eight electrons.
* Central bonding atoms from third period may carry more than eight electrons because of empty d-orbitals.
6. If more than one possible structures exist, calculate the formal charge on each bonded atoms.
* Formal charge = (Group number) - (No. of unshared electrons + No. of electron-pair bonds) or
* Formal charge = [total number of valence electrons in the free atom] - [Total number of lone pari electrons] - (1/2)[Total
number of electron-pair bonds]
* Structure with formal charge of 0 on each atom is preferred. If formal charge of 0 is not possible, a structure with negative charge
on the most electronegative atom and positive charge on the most electropositive element is preferred.
* The sum of formal charges of all atoms in a given molecule or ion must equal the overall charge on that species.
2. Choose a central bonding atom from the given molecule.
* Central bonding atom is the least electronegative atom.
* Exceptions: Hydrogen (H) and Fluorine (F) always occupy peripheral position.
3. Draw a single sigma bond (-) between the central bonding atom and each peripheral atom.
4. Draw three unshared electron pairs, called lone-pairs, to complete octet on each peripheral atom except for Hydrogen (H), which
follows duet rule.
* If extra electrons remain, place them on the central bonding atom.
5. Complete octet for the central bonding atom by moving lone pairs from peripheral atoms.
* Atoms with less than four valence electrons like Li, Be, and B have less than eight electrons.
* Central bonding atoms from third period may carry more than eight electrons because of empty d-orbitals.
6. If more than one possible structures exist, calculate the formal charge on each bonded atoms.
* Formal charge = (Group number) - (No. of unshared electrons + No. of electron-pair bonds) or
* Formal charge = [total number of valence electrons in the free atom] - [Total number of lone pari electrons] - (1/2)[Total
number of electron-pair bonds]
* Structure with formal charge of 0 on each atom is preferred. If formal charge of 0 is not possible, a structure with negative charge
on the most electronegative atom and positive charge on the most electropositive element is preferred.
* The sum of formal charges of all atoms in a given molecule or ion must equal the overall charge on that species.
Exceptions of Lewis Structure to the octet rule:
1. Incomplete octet of central bonding atom.
As pointed out earlier in step 5, central bonding atoms like Lithium (Li), Beryllium (Be) and Boron (B) which have less than 4 valence electrons less than 8
electrons, and therefore contradicts octet rule.
As pointed out earlier in step 5, central bonding atoms like Lithium (Li), Beryllium (Be) and Boron (B) which have less than 4 valence electrons less than 8
electrons, and therefore contradicts octet rule.
2. The expanded octet.
Elements in and beyond the third period of periodic table may have more than eight electrons when they are the central bonding atom.
Elements in and beyond the third period of periodic table may have more than eight electrons when they are the central bonding atom.
3.Lewis structures with odd-electron:
when the total number of valence electrons in a molecule is odd, octet is disobeyed.
when the total number of valence electrons in a molecule is odd, octet is disobeyed.
2. The Valence Shell Electron Pair Repulsion (VSEPR) Theory:
As pointed out earlier, Lewis dot structure fails to provide the shapes of covalent molecule. The VSEPR Theory, on the other hand, helps us to predict the shapes of covalent molecule.
It is important to note in the VSEPR theory that
* Multiple bonds (double or triple bonds) count as one effective pair.
* For a molecule with more than one structure, i.e. if the molecule exhibits resonance, use any one of the resonance structures can be
used to predict the molecular structure.
* Electron pairs in the valence shell repel one another because of negative charge and the repulsive interaction decreases in the order
shown below:
Lone pair (lp) - Lone pair (lp) > Lone pair (lp) - Bond pair (bp) > Bond pair (bp) - Bond pair (bp).
It is important to note in the VSEPR theory that
* Multiple bonds (double or triple bonds) count as one effective pair.
* For a molecule with more than one structure, i.e. if the molecule exhibits resonance, use any one of the resonance structures can be
used to predict the molecular structure.
* Electron pairs in the valence shell repel one another because of negative charge and the repulsive interaction decreases in the order
shown below:
Lone pair (lp) - Lone pair (lp) > Lone pair (lp) - Bond pair (bp) > Bond pair (bp) - Bond pair (bp).
Steps for using VSEPR model to predict molecular structure:
1. Draw Lewis structure for the molecule.
2. Count the electron pairs (bonding and lone-pairs) around the central bonding atom.
* remember that multiple bond (double bond or triple bond) count as one bonding pair.
3. Use the number of electron pairs and table 1 or 2 to determine the arrangement of electron pairs.
4. Name the molecular structures from the position of atoms not the electron pairs.
2. Count the electron pairs (bonding and lone-pairs) around the central bonding atom.
* remember that multiple bond (double bond or triple bond) count as one bonding pair.
3. Use the number of electron pairs and table 1 or 2 to determine the arrangement of electron pairs.
4. Name the molecular structures from the position of atoms not the electron pairs.
Geometries of molecules from VSEPR Theory:
1. Geometries of molecules without lone pair(s) on the central bonding atom are shown in the table below:
2. Geometries of molecules with one or more lone pairs on the central bonding atom are shown in the table below:
3. Hybridization:
Hybridization is the mixing of native atomic orbitals of nearly similar energies to form a new set of orbitals (Hybridized orbitals) of similar energies for bonding.
Features of hybridization:
1. The number of hybridized orbitals is equal to the total number of orbitals from which it is formed. In figure 1, one 2s orbital and three 2p orbitals hybridize to
form four sp3 hybridized orbitals.
2. Hybridized orbitals have same energy and shape.
In figure 1, all the four sp3 hybridized orbitals have the same energy which is greater than that of 2s but less than that of 3p.
3. Hybridized orbitals are oriented in such a way that there is minimum electron pair repulsion which gives us the geometric shape(s) of a molecule.
form four sp3 hybridized orbitals.
2. Hybridized orbitals have same energy and shape.
In figure 1, all the four sp3 hybridized orbitals have the same energy which is greater than that of 2s but less than that of 3p.
3. Hybridized orbitals are oriented in such a way that there is minimum electron pair repulsion which gives us the geometric shape(s) of a molecule.
Types of Hybridization:
There are different types of hybridization involving s, p, and d orbitals.
a. sp hybridization: In sp hybridization, one s orbital (spherical shape) and one p orbital (dumbbell shape) of nearly same but different energy combine to form
two sp hybridized orbitals of same energy and shape.
Example of molecules with sp hybridized orbital: BeCl2, CO, N2, C2H2
b. sp2 hybridization: In sp2 hybridization, one s orbital and two p orbitals of nearly same but different energy combine to form three sp2 hybridized orbitals of
same energy and shape.
Example of a molecules with sp2 hybridization: BCl3, C2H4
c. sp3 hybridization: In sp3 hybridization, one s orbital and three p orbitals of nearly same but different energy combine to form four sp3 hybridized
orbitals of same energy and shape.
Example of molecules with sp3 hybridization: NH3, H2O, C2H6
d. sp3d or dsp3 hybridization: In sp3d and dsp3 hybridization, one s orbital, three p orbitals and one d orbital of nearly same but different energy combine to
form 5 sp3d or dsp3 hybridized orbitals of same energy and shape.
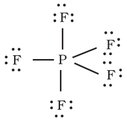
Example of molecules with sp3d and dsp3 hybridization: PF5 (sp3d hybridized) and BrF5 (dsp3 hybridized).
e. sp3d2 or d2sp3 hybridization: In sp3d2 or d2sp3 hybridization, one s orbital, 3 p orbitals, and 2 d orbitals of nearly same but different energy combine to form
six sp3d2 or d2sp3 hybridized orbitals of same energy and shape.
Example of molecules with sp3d2 and d2sp3 hybridization: SF6 (sp3d2 hybridized) and XeF4 (d2sp3 hybridized).
a. sp hybridization: In sp hybridization, one s orbital (spherical shape) and one p orbital (dumbbell shape) of nearly same but different energy combine to form
two sp hybridized orbitals of same energy and shape.
Example of molecules with sp hybridized orbital: BeCl2, CO, N2, C2H2
b. sp2 hybridization: In sp2 hybridization, one s orbital and two p orbitals of nearly same but different energy combine to form three sp2 hybridized orbitals of
same energy and shape.
Example of a molecules with sp2 hybridization: BCl3, C2H4
c. sp3 hybridization: In sp3 hybridization, one s orbital and three p orbitals of nearly same but different energy combine to form four sp3 hybridized
orbitals of same energy and shape.
Example of molecules with sp3 hybridization: NH3, H2O, C2H6
d. sp3d or dsp3 hybridization: In sp3d and dsp3 hybridization, one s orbital, three p orbitals and one d orbital of nearly same but different energy combine to
form 5 sp3d or dsp3 hybridized orbitals of same energy and shape.
Example of molecules with sp3d and dsp3 hybridization: PF5 (sp3d hybridized) and BrF5 (dsp3 hybridized).
e. sp3d2 or d2sp3 hybridization: In sp3d2 or d2sp3 hybridization, one s orbital, 3 p orbitals, and 2 d orbitals of nearly same but different energy combine to form
six sp3d2 or d2sp3 hybridized orbitals of same energy and shape.
Example of molecules with sp3d2 and d2sp3 hybridization: SF6 (sp3d2 hybridized) and XeF4 (d2sp3 hybridized).
Types of Covalent Bond
Covalent bond can be divided into two types: 1. Sigma (σ) bond and 2. pi (⫪) bond.
1. Sigma (σ) bond is formed from hybrid orbitals whose lobes point toward each other
2. pi (⫪) bond is formed from overlapping of parallel p - orbitals.
1. Sigma (σ) bond is formed from hybrid orbitals whose lobes point toward each other
2. pi (⫪) bond is formed from overlapping of parallel p - orbitals.
|
|
|
HTML Comment Box is loading comments...