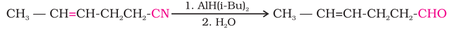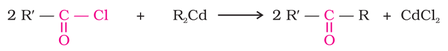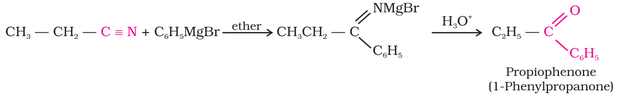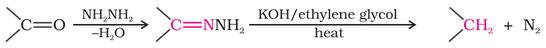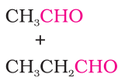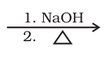Aldehydes, Ketones and Carboxylic acids
IUPAC Nomenclature of Aldehydes:
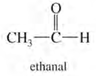
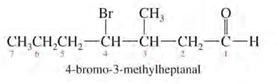
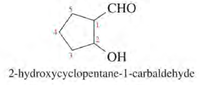
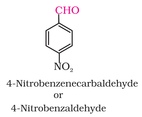
1. Number the longest chain starting from carbon of aldehyde group.
2. Name the compound by changing -e to -al (e.g. ethane to ethanal).
Examples:
2. Name the compound by changing -e to -al (e.g. ethane to ethanal).
Examples:
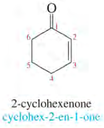
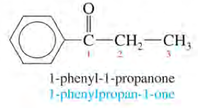
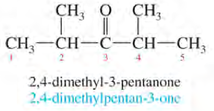
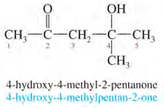
IUPAC Nomenclature of ketones:
1. Number the longest chain from the end nearest to the carbonyl group.
2. Name the compound by changing -e to -one (e.g. ethane to ethanone).
Examples:
2. Name the compound by changing -e to -one (e.g. ethane to ethanone).
Examples:
Preparation of Aldehydes:
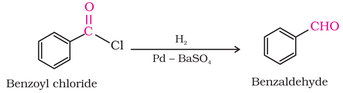
1. From acyl chloride (Rosenmund reduction) :
2. From nitriles:
(I) Stephen reaction :
(I) Stephen reaction :
(II) reduction of nitrile by diisobutylaluminium hydride ( AlH(i-Bu)2) and (DIBAL-H) to imine followed by hydrolysis:
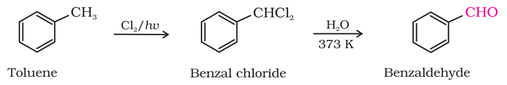
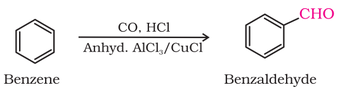
3. From aromatic hydrocarbons to aromatic aldehydes:
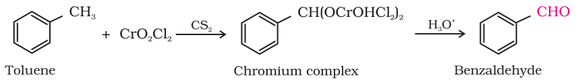
(I) By oxidation of methylbenzene:
(a) Using chromyl chloride (CrO2Cl2) (Etard reaction):
(a) Using chromyl chloride (CrO2Cl2) (Etard reaction):
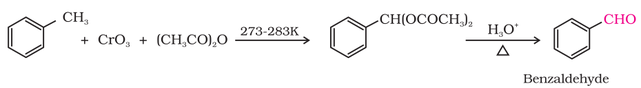
(b) Using chromic oxide (CrO3):
(II) Chlorination followed by hydrolysis:
(III) by Gatterman - Koch reaction:
Preparation of Ketones:
1. From acyl chlorides:
2. From nitriles:
3. From Friedel-Crafts acylation reaction:
Reactions of aldehydes and ketones:
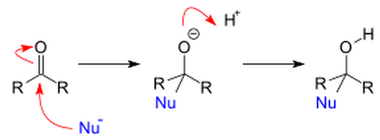
Aldehydes and ketones undergo nucleophilic substitution reaction. In nucleophilic substitution, a species that has extra electron or lone pair of electron attacks the carbonyl carbon of the ketone or the aldehyde leading to an alkoxide intermediate as shown in the middle figure below. This alkoxide intermediate can capture a proton to produce a neutral species as shown below.
Reactivity of aldehyde vs ketone:
Aldehydes are usually more reactive than Ketones for two reasons: 1. steric and 2 electronic reasons
Sterically, the presence of two bulky (large) groups in ketones will hinder the attack of nucleophile to carbonyl carbon in ketone. Aldehydes have only one bulky group around the carbonyl carbon and it is easier for the nucleophile to attack the carbonyl carbon as compared to ketones.
Electronically, aldehyde is also more reactive than ketone because the presence of two alkyl groups in ketones will reduce the electrophilicity (partial positive charge ) of the carbonyl carbon due to inductive effect by the alkyl groups. Therefore, attack of nucleophile on carbonyl carbon in ketone is harder than attack of carbonyl carbon in aldehyde making the aldehyde more reactive.
Aldehydes are usually more reactive than Ketones for two reasons: 1. steric and 2 electronic reasons
Sterically, the presence of two bulky (large) groups in ketones will hinder the attack of nucleophile to carbonyl carbon in ketone. Aldehydes have only one bulky group around the carbonyl carbon and it is easier for the nucleophile to attack the carbonyl carbon as compared to ketones.
Electronically, aldehyde is also more reactive than ketone because the presence of two alkyl groups in ketones will reduce the electrophilicity (partial positive charge ) of the carbonyl carbon due to inductive effect by the alkyl groups. Therefore, attack of nucleophile on carbonyl carbon in ketone is harder than attack of carbonyl carbon in aldehyde making the aldehyde more reactive.
2. Reduction:
(i) Reduction of aldehydes and ketones to primary or secondary alcohol using sodium borohydride or lithium aluminum hydride.
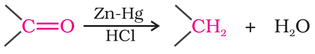
(ii) Reduction of aldehydes or ketones to hydrocarbons using Clemmenson reduction or Wolff-Kishner reduction
3. Oxidation:
Aldehydes can be easily oxidized to carboxylic acids using nitric acid, potassium permanganate, potassium dichromate, etc. Even mild oxidizing agents, mainly Tollens’ reagent and Fehlings’ reagent also oxidize aldehydes.
Oxidation of ketones, however, require more stronger oxidizing agent in more vigorous conditions.
Oxidation of ketones, however, require more stronger oxidizing agent in more vigorous conditions.
(i) Tollen's test (used to distinguish aldehydes from ketones, aldehydes give positive test by producing a bright silver mirror):
(ii) Fehling's test (used to distinguish aldehydes from ketones, aldehydes give positive test by producing a red-brown ppt. except
aromatic aldehydes):
aromatic aldehydes):
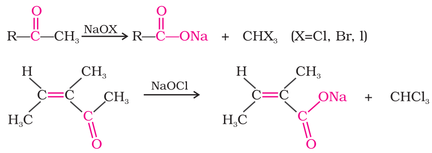
(iii) Oxidation of methyl ketones using haloform reaction: both aldehydes and ketones having at least one methyl group
attached to the carbonyl carbon will react to give a product having one less carbon atom as shown in the reaction below:
attached to the carbonyl carbon will react to give a product having one less carbon atom as shown in the reaction below:
4. Reaction due to α-hydrogen:
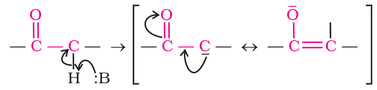
Aldehydes and ketones also undergo reaction due to acidic nature of alpha hydrogen atom. The acidic nature of alpha hydrogen of aldehydes and ketones is due to strong electron withdrawing nature of carbonyl group and resonance stabilization of conjugate base as shown below:
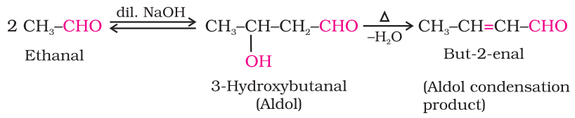
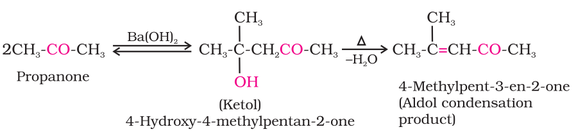
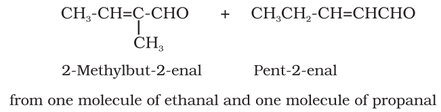
(i) Aldol condensation: Aldehydes and ketones having at least one hydrogen α-hydrogen undergoes reaction in weakly
basic condition to form β-hydroxy aldehydes (aldol) or β-hydroxy ketones (ketol), respectively. β-hydroxy aldehydes (aldol) or β
hydroxy ketones (ketol) readily loses water to give α,β-unsaturated carbonyl compounds.
basic condition to form β-hydroxy aldehydes (aldol) or β-hydroxy ketones (ketol), respectively. β-hydroxy aldehydes (aldol) or β
hydroxy ketones (ketol) readily loses water to give α,β-unsaturated carbonyl compounds.
2. Cross aldol condensation: When aldol condensation is carried out between two different aldehydes or ketones or an aldehyde
and a ketone having alpha-hydrogen, the reaction is called cross cross aldol condensation.
and a ketone having alpha-hydrogen, the reaction is called cross cross aldol condensation.
Other reactions:
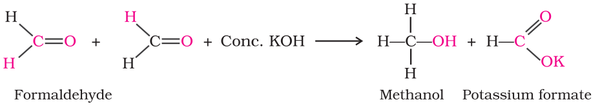
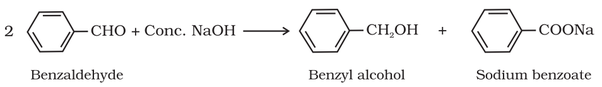
(i) Cannizzaro reaction: Aldehydes with not alpha-hydrogen is self oxidized and reduced in the presence of conc. base to form
alcohol and carboxylic acid salt respectively.
alcohol and carboxylic acid salt respectively.
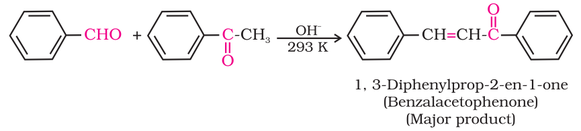
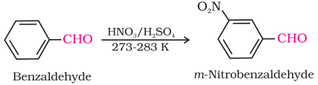
(ii) Electrophilic substitution reaction: Aromatic aldehydes and ketones act as moderately deactivating group and electrophiles
are added to the meta position.
are added to the meta position.