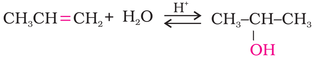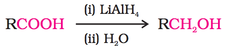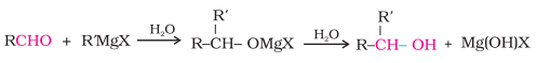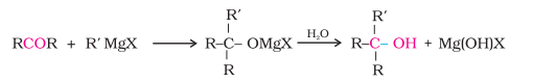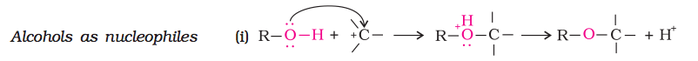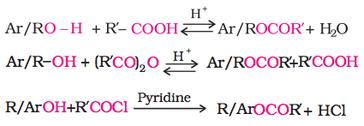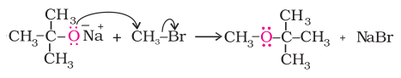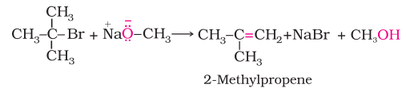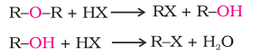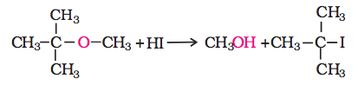Alcohols, Phenols, and Ethers
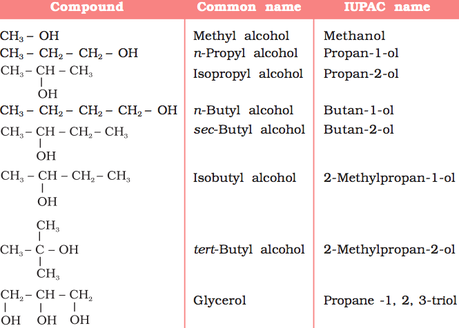
IUPAC Nomenclature of alcohol
1. Number the longest carbon chain.
2. Name the chain by replacing -e to -ol, make sure to number the number of C on which the alcohol group is attached.
3. If multiple alcohol groups are present, don't replace -e but use -diol or -triol as shown in the last row below.
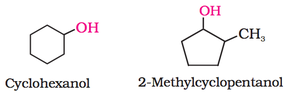
3. If the alcohol is cyclic, use cyclo as shown below.
2. Name the chain by replacing -e to -ol, make sure to number the number of C on which the alcohol group is attached.
3. If multiple alcohol groups are present, don't replace -e but use -diol or -triol as shown in the last row below.
3. If the alcohol is cyclic, use cyclo as shown below.
Structures of alcohols, phenols, and ethers
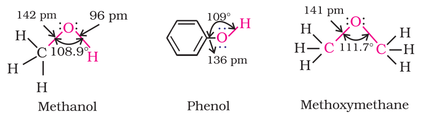
Structures of methanol, phenol, and methoxymethane is given below:
Alcohol: The bond angle of alcohol between C-O-H shown above is slightly less than tetrahedral angle (109.5 degree) because of repulsion between lone pair electrons of oxygen atom as shown in the figure of methanol.
Phenol: The carbon - oxygen bond length (136 pm) of phenol, as shown above, is slightly less than that of methanol and methoxymethane due to (i) conjugation of unshared electron pair of oxygen with the aromatic ring and (ii) sp2 hybridised carbon to with oxygen is attached.
Ether: Even though ether is tetrahedral in structure, the bond angle is slightly greater than tetrahedral angle because of repulsion between two bulky groups attached to the oxygen atom.
Phenol: The carbon - oxygen bond length (136 pm) of phenol, as shown above, is slightly less than that of methanol and methoxymethane due to (i) conjugation of unshared electron pair of oxygen with the aromatic ring and (ii) sp2 hybridised carbon to with oxygen is attached.
Ether: Even though ether is tetrahedral in structure, the bond angle is slightly greater than tetrahedral angle because of repulsion between two bulky groups attached to the oxygen atom.
Physical Properties of alcohols and phenols:
Boiling point: The boiling points of alcohols and phenols increase with increase in the number of carbon atoms because of increase in van der Waals forces with increase in surface area. In alcohols, the boiling points decrease with increase of branching in carbon chain because of decrease in van der Waals forces with decrease in surface area.
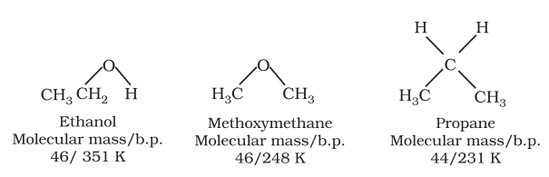
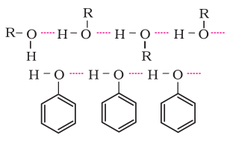
Comparison of boiling points: The boiling points of alcohols and phenols are higher in comparison to other classes of compounds such as hydrocarbons, ethers, haloalkanes and haloarenes of comparable molecular masses. The high boiling points of alcohols are mainly due to the presence of intermolecular hydrogen bonding in them which is lacking in hydrocarbons, ethers, haloalkanes and haloarenes.
Comparison of boiling points: The boiling points of alcohols and phenols are higher in comparison to other classes of compounds such as hydrocarbons, ethers, haloalkanes and haloarenes of comparable molecular masses. The high boiling points of alcohols are mainly due to the presence of intermolecular hydrogen bonding in them which is lacking in hydrocarbons, ethers, haloalkanes and haloarenes.
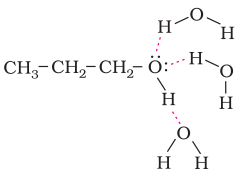
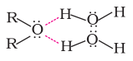
Solubility: Alcohols and phenols are soluble in water due to their ability to form hydrogen bond with water molecule as shown below. The solubility decreases with increase in the size of alkyl or aryl group due to their hydropho-phobic nature.
Preparation of Alcohols
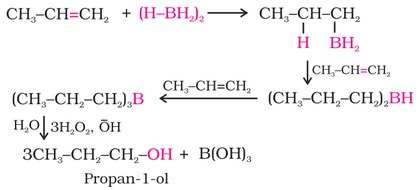
1. From alkenes
|
|
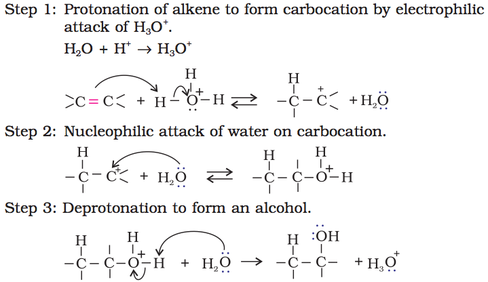
(i) Acid catalysed hydration:
(ii) By Hydroboration-Oxidation:
|
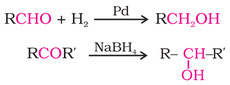
2. From Carbonyl Compounds:
3. From Grignard reagents :
Preparation of Phenols
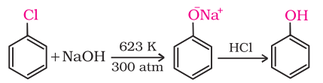
1. From haloarenes:
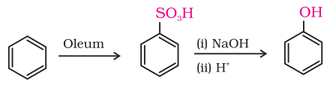
2. From benzenesulphonic acid:
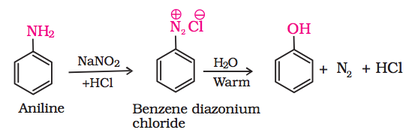
3. From diazonium salts:
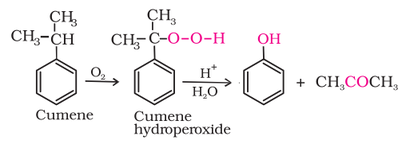
4. From cumene:
Reaction of alcohols
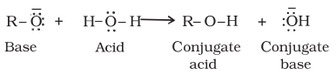
Alcohols can react as either nucleophile or as electrophile. When alcohol reacts as nucleophile, the Oxygen attacks another atom such as Carbon, eventually losing the Hydrogen atom attached to the Oxygen. Therefore in a nucleophilic attack of alcohol, O-H bond is broken as shown below.
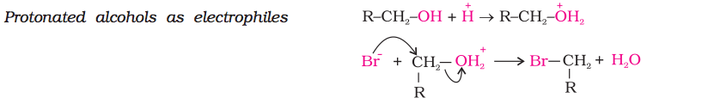
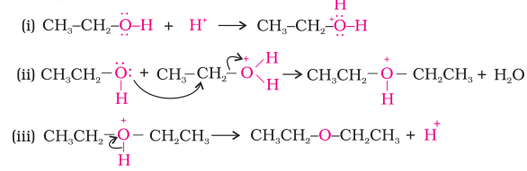
When alcohol reacts as electrophile, another electronegative atom attacks the C atom adjacent to the protonated O atom leading to the leaving of protonated -OH2+ group as shown in the reaction below. Therefore in an electrophilic reaction of alcohol, C-O bond is broken.
(a) Reactions involving cleavage of O-H bond (nucleophilic):
1. Acidity of alcohols and phenols
|
|
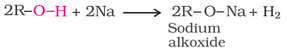
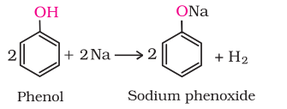
(i) Reaction with metals:
(ii) Acidity of alcohol:
(iii) Acidity of phenol:
|
2. Esterification
(b) Reactions involving cleavage of carbon - oxygen (C-O) bond in alcohols (electrophilic reaction):
1. Reaction with hydrogen halides (alcohol to alkyl halide):
|
|
Alcohols reacts with hydrogen halides to form alkylhalides. R-OH + H-X -------------> R-X + H2O, where X is any halide.
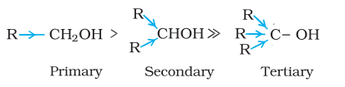
This reaction is also used to identify if a given sample is tertiary, secondary, or primary alcohol by using HCL. This test is called the Lucas test. Tertiary halides produce a cloudy product immediately after adding Lucas reagent (HCl + ZnCl2) while primary alcohol do not produce any cloudy product at room temperature. |
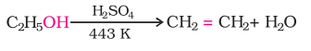
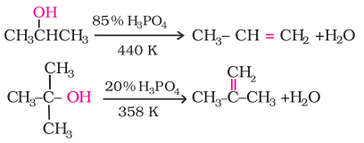
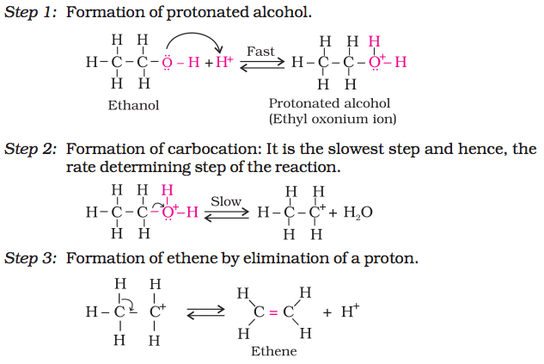
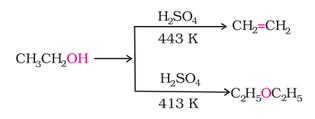
2. Dehydration (Alcohol to alkene):
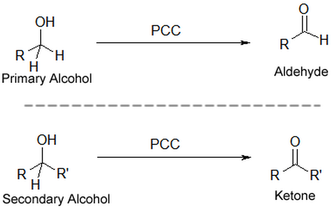
3. Oxidation (Alcohol to aldehyde and ketones):
(c) Reaction of Phenols:
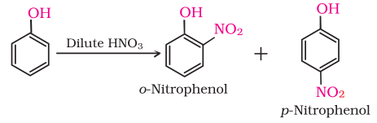
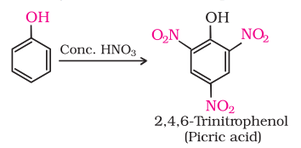
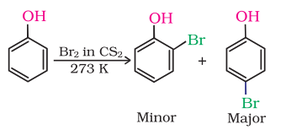
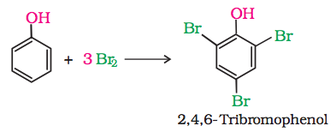
1. Electrophilic aromatic substitution
|
|
|
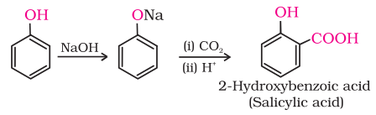
2. Kolbe’s reaction
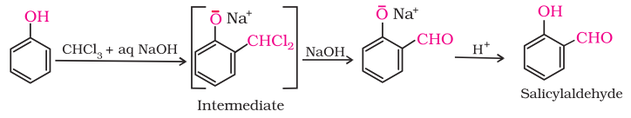
3. Reimer-Tiemann reaction
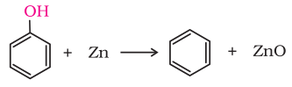
4. Reaction of phenol with zinc dust
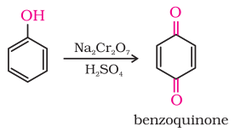
5. Oxidation
Physical Properties of ethers:
Boiling points: Boiling points of ethers are similar to the alkanes of similar molecular masses. However, the boiling point of ethers are much lower than the boiling points of alcohols of similar molecular masses due to the presence of hydrogen bonding in alcohols.
Miscibility: The miscibility of ethers with water resembles those of alcohols of the same molecular mass. This is due to the ability of oxygen in ether to form hydrogen bonds with water molecules like alcohols.
Preparation of ethers:
1. By dehydration of alcohols:
2. Williamson systhesis:
|
|
Williamson ether synthesis is used for the formation of ethers from alkyl halide and sodium alkoxide as shown in the reaction below.
Better results are obtained if the alkyl halide is primary. In case of secondary and tertiary alkyl halides, elimination competes over substitution. If a tertiary alkyl halide is used, an alkene is the only reaction product and no ether is formed. For example, the reaction of CH3ONa with (CH3)3C–Br gives exclusively 2-methylpropene.
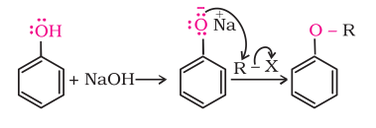
Phenols are also converted to ether by first reacting with NaOH, and then the desired alkyl halide as shown below.
|
Reactions of ethers:
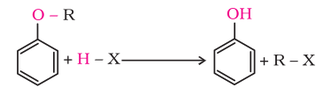
1. Cleavage of C–O bond in ethers
|
|
The reaction of dialkyl ether gives two alkyl halide molecules.
Alkyl aryl ethers are cleaved at the alkyl-oxygen bond due to the more stable aryl-oxygen bond. The reaction yields phenol and alkyl halide.
Reaction Mechanism:
However, when one of the alkyl group is a tertiary group, the halide formed is a tertiary halide as shown below. It is because in step 2 of the reaction, the departure of leaving group (HO–CH3) creates a more stable carbocation [(CH3)3C+], and the reaction follows SN1 mechanism.
|
1. Electrophilic substitution
|
|
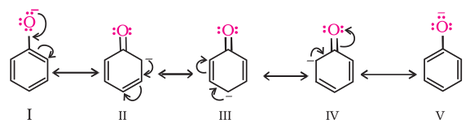
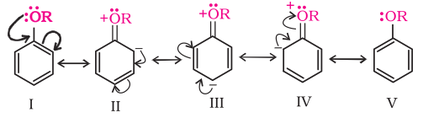
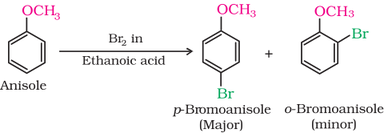
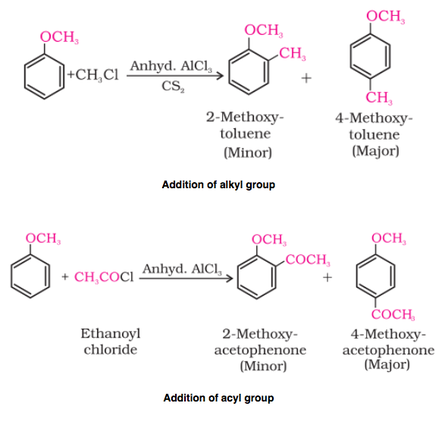
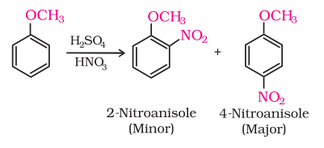
The alkoxy group (-OR) is ortho, para directing and activates the aromatic ring towards electrophilic substitution in the same way as in phenol. The ortho, and para directing nature can be seen from it's resonance structure as shown below.
(i) Halogenation (addition of halogen):
(ii) Friedel-Crafts reaction (addition of alkyl and acyl group):
(iii) Nitration:
|
HTML Comment Box is loading comments...